Optical single-channel recording by imaging Ca2+ flux through individual ion channels: theoretical considerations and limits to resolution
Abstract
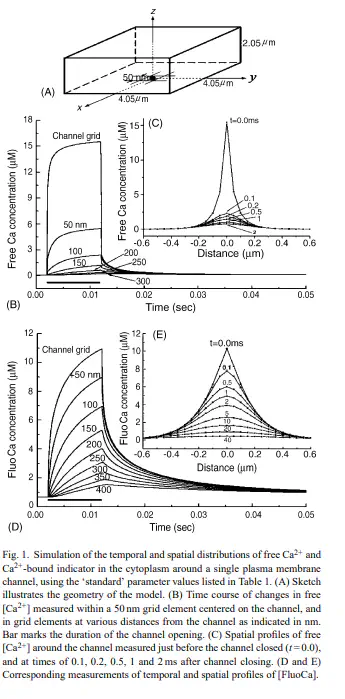
Recent developments in microscopy and fluorescent indicators now make it possible to monitor the activity and localization of membrane ion channels by imaging Ca(2+) flux through individual channels. Such optical approaches have advantages over electrophysiological single-channel techniques in that they are less invasive, provide spatial information and can simultaneously and independently monitor hundreds of channels. However, their kinetic resolution does not yet approach that of patch-clamp recordings. To help understand the processes that determine the temporal resolution and noise level of single-channel Ca(2+) fluorescence signals (SCCaFTs), we simulated the microdomains of Ca(2+) ions and Ca(2+)-bound indicator dye that exist around the mouth of an open channel. Further, as an aid to development of improved optical techniques, we modeled the dependence of the amplitude and kinetics of SCCaFTs on parameters such as the imaging volume, the indicator concentration, affinity and mobility, and the presence of endogenous and exogenous Ca(2+) buffers. The results indicate that under optimal conditions, including the use of confocal or total-internal reflection microscopy to image from sub-femtolitre volumes, SCCaFTs should resolve channel openings as brief as 1ms with a signal-to-noise ratio >10.